- What is chemical equilibrium?
- What is weak alkali?
- What is the air pressure on the earth’s surface?
- What is the activity of an element?
- Calcium carbonate Uses, Benefits, and Side Effects
Why is Calcium called alkaline earth metal?
Calcium is called an alkaline earth metal because it is a member of Group 2A (or IIA) of the periodic table. The elements in this group are all characterized by two valence electrons in their outermost shells, which they
can easily lose to form cations with a +2 charge. This is why the alkaline earth metals are all relatively reactive metals.
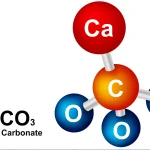
The term “alkaline earth” comes from the fact that the oxides of alkaline earth metals are basic (alkaline) and were originally thought to be “earth” (nonmetallic substances). When the oxides of the alkaline earth metals are dissolved in water, they form hydroxide solutions, which are bases. For example, calcium oxide (CaO) reacts with water to form calcium hydroxide (Ca(OH)2), which is a strong base.
The alkaline earth metals are important elements in many biological and industrial processes. Calcium, for example, is essential for bone health and muscle function. It is also used in the production of cement, plaster, and other building materials. Magnesium is another important alkaline earth metal. It is essential for photosynthesis and is used in many alloys and other materials.
Here is a summary of the reasons why calcium is called an alkaline earth metal:
- It is a member of Group 2A (or IIA) of the periodic table, which is the group of alkaline earth metals.
- It has two valence electrons in its outermost shell, which it can easily lose to form cations with a +2 charge.
- Its oxide (calcium oxide, CaO) is basic (alkaline) and was originally thought to be an “earth.”
- Its oxide reacts with water to form a hydroxide solution (calcium hydroxide, Ca(OH)2), which is a base.